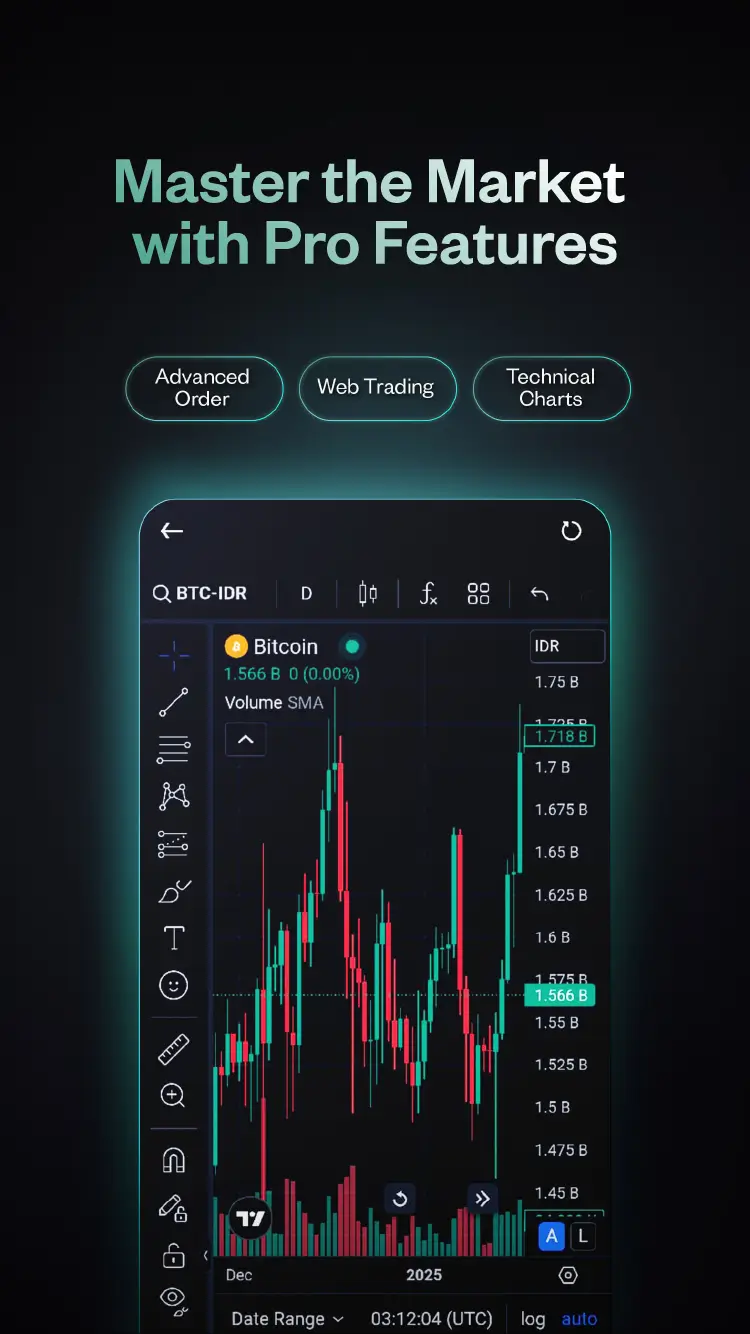
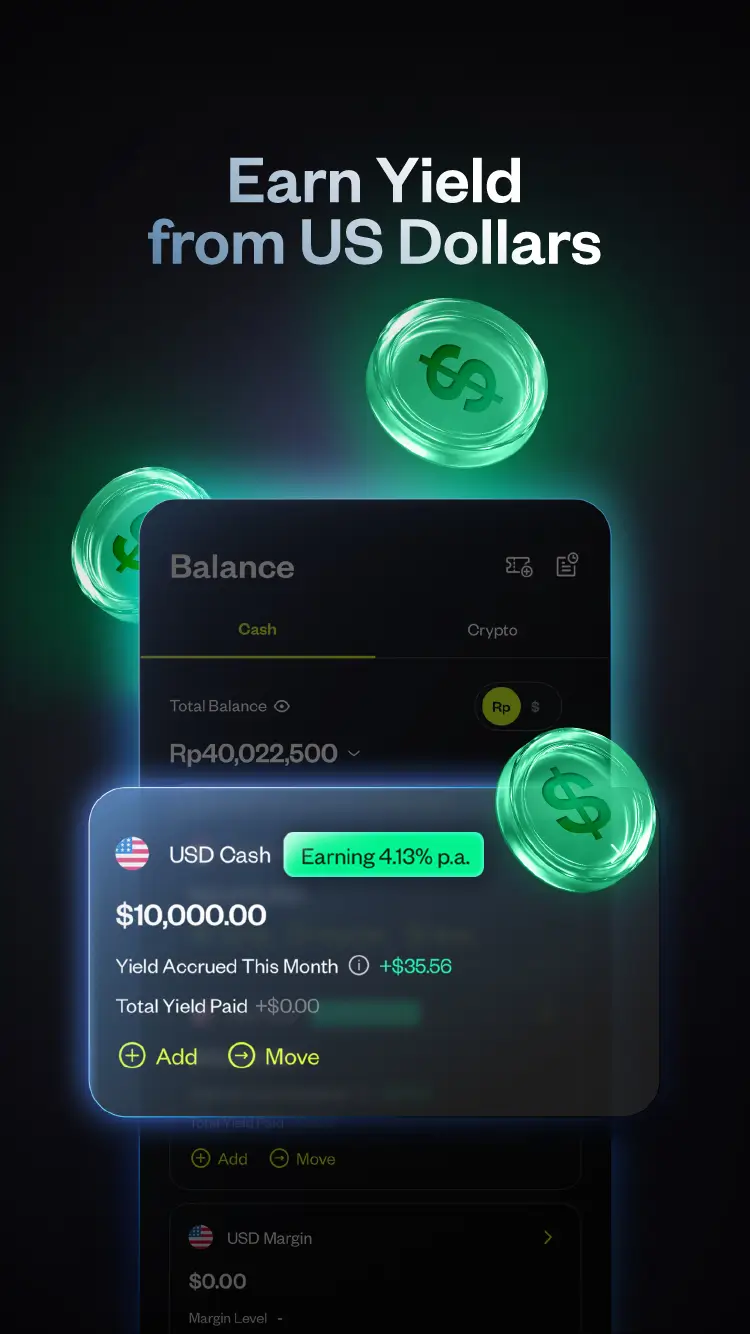
Trade on Pluang
One platform for all markets
Download
Oculis secures FDA agreement on trial design for Privosegtor in optic neuritis treatment

Oculis Holding AG has received FDA Special Protocol Assessment (SPA) approval for its Phase 3 PIONEER-1 trial of Privosegtor, a novel neuroprotective drug for optic neuritis (ON). This agreement confirms the trial design is suitable to support a future New Drug Application if results are positive. Privosegtor has shown promise in earlier trials and holds FDA Breakthrough Therapy and EMA PRIME designations, highlighting its potential to address a significant unmet need in neuro-ophthalmology. The PIONEER-1 trial will assess visual improvement and long-term safety in patients, including those with multiple sclerosis, aiming to offer the first neuroprotective therapy for ON.