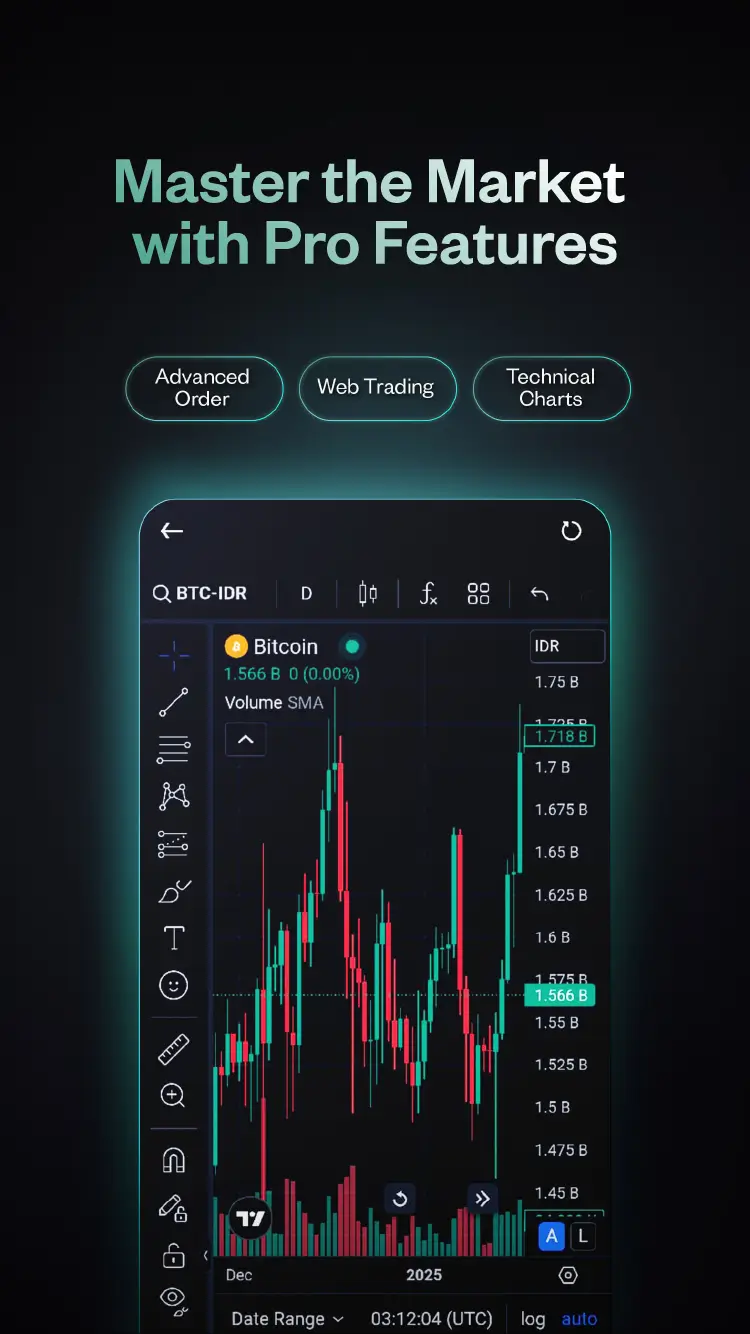
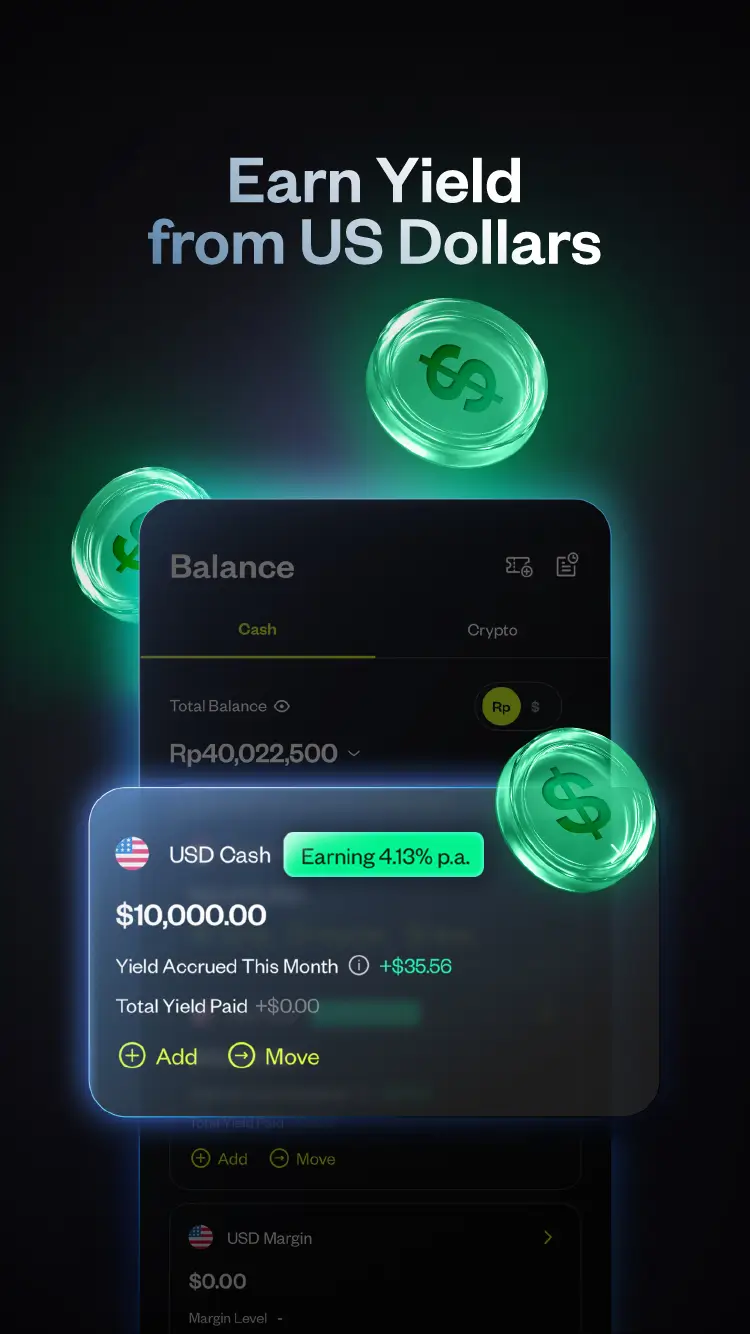
Trade on Pluang
One platform for all markets
Download
Bluejay Diagnostics advances clinical trials and manufacturing for sepsis test in Q1 2026.

Bluejay Diagnostics reported progress in Q1 2026 on its Symphony IL-6 test for sepsis monitoring, enrolling about 680 of 750 patients in a pivotal clinical study. The company resolved manufacturing issues with its Symphony cartridges and is preparing for FDA regulatory submission. With $3.7 million cash and a $1.9 million net loss, Bluejay focuses on clinical, manufacturing, and regulatory milestones to enable rapid near-patient testing for critical care. The next steps include completing validation and pursuing commercialization.