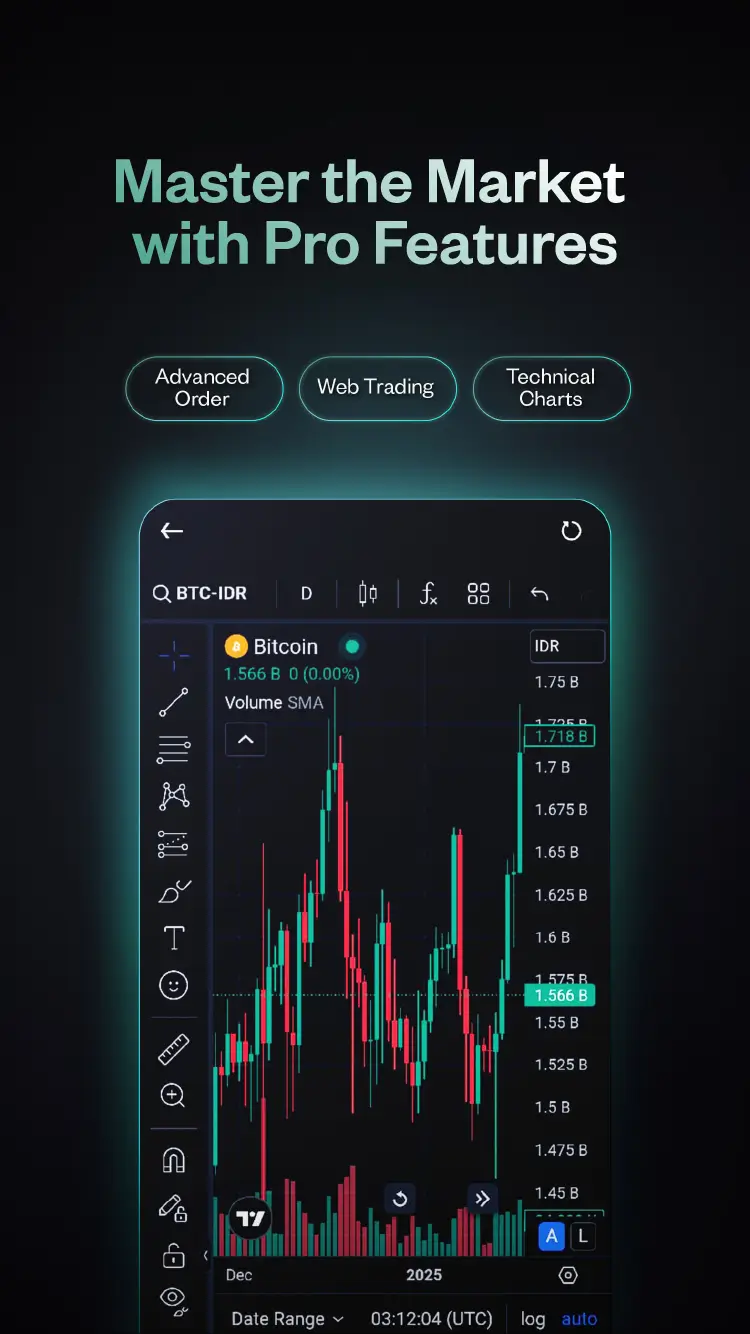
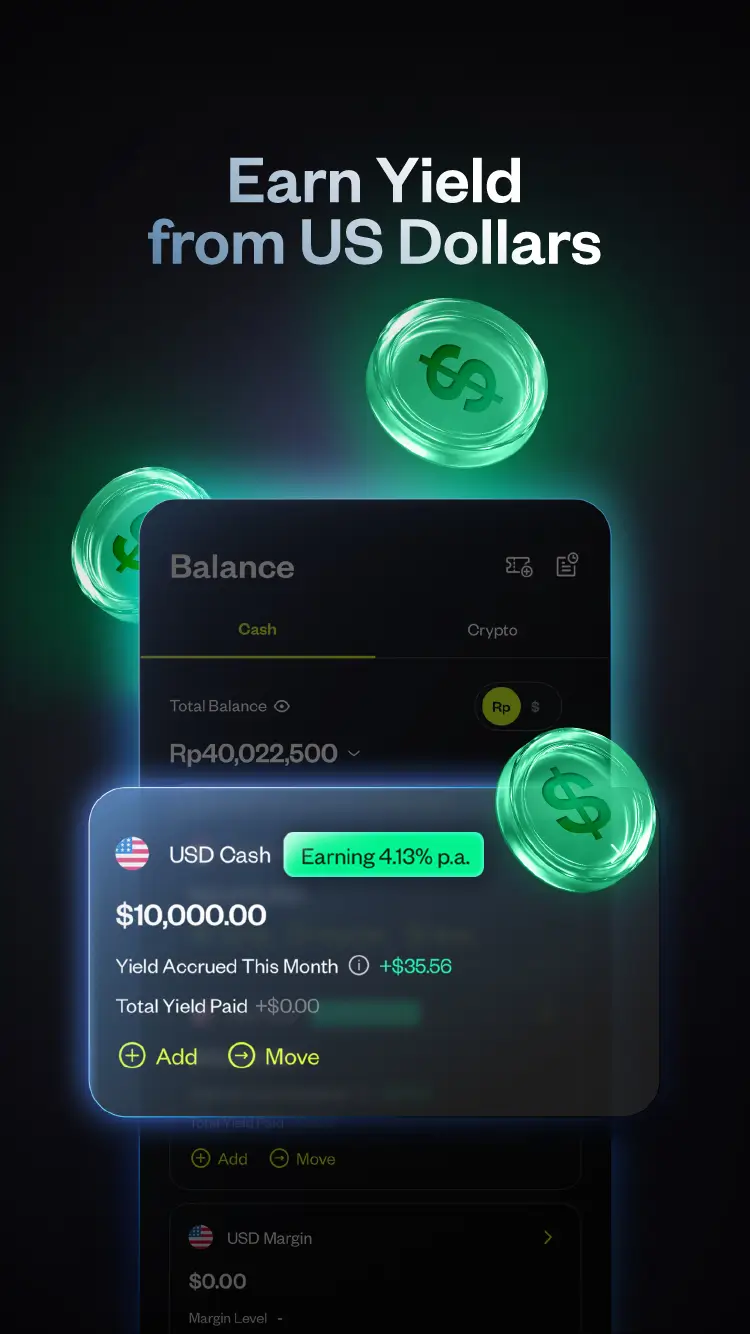
Trade on Pluang
One platform for all markets
Download
FDA approves VYVGART for all adult generalized myasthenia gravis patients, expanding treatment options.

The U.S. FDA has approved an expanded label for argenx's VYVGART and VYVGART Hytrulo to treat all adult patients with generalized myasthenia gravis (gMG), including all serotypes such as anti-AChR-Ab positive, anti-MuSK-Ab positive, anti-LRP4-Ab positive, and triple seronegative. This approval is based on the Phase 3 ADAPT SERON study, which showed rapid, significant, and sustained symptom improvements across these patient groups. The expanded indication simplifies treatment decisions and addresses a previously underserved patient population, offering broader access to this targeted therapy. argenx continues to develop VYVGART for additional MG populations, including pediatric patients.