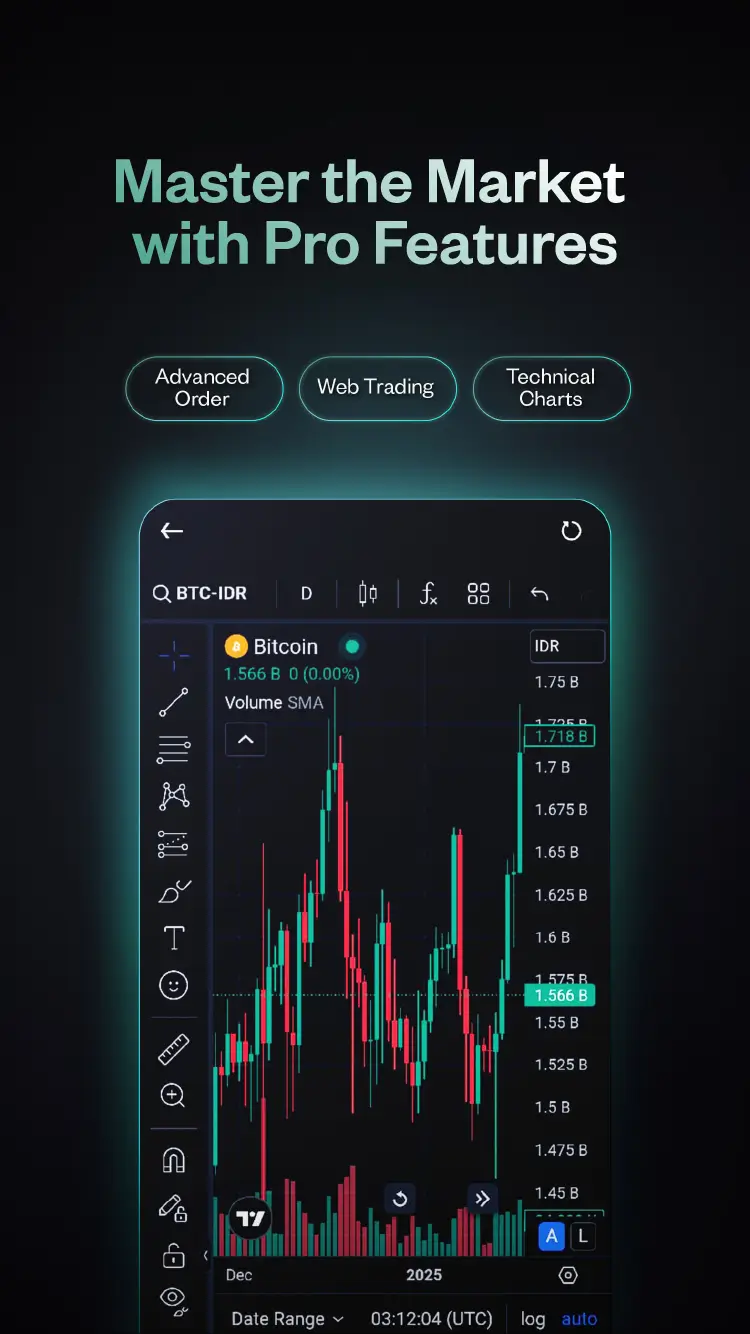
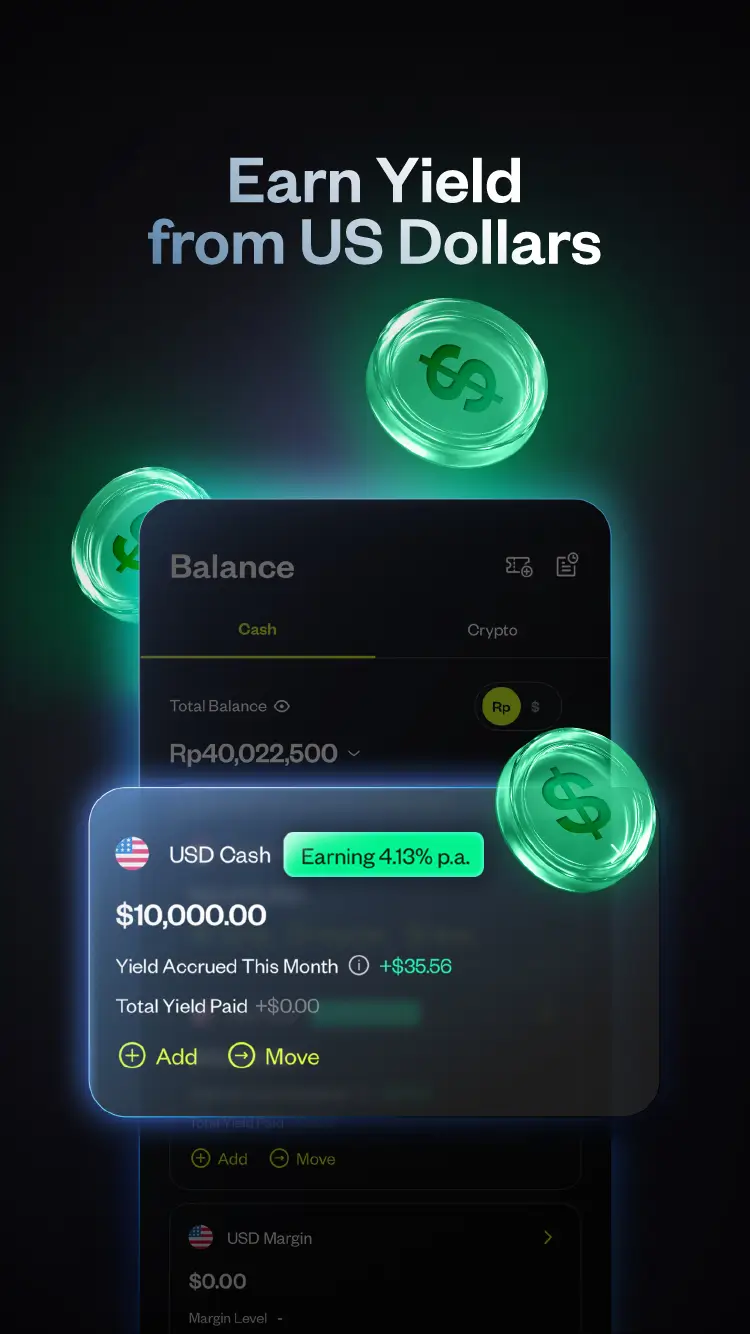
Trade on Pluang
One platform for all markets
Download
FDA completes inspection at Alvotech's Reykjavik facility; company on track for BLA resubmissions this quarter

Alvotech announced that the FDA has completed its routine cGMP surveillance inspection at the company's manufacturing site in Reykjavik, Iceland, issuing a Form 483 with observations the company believes can be quickly addressed. This positive outcome supports Alvotech's plan to resubmit Biologics License Applications (BLAs) in the second quarter of 2026. The company remains confident in the strong compliance and improvements at the site and expects FDA approval of the relevant BLAs within the year. This progress is crucial for Alvotech's continued development and commercialization of biosimilar medicines globally.